
Research
• Preface
•
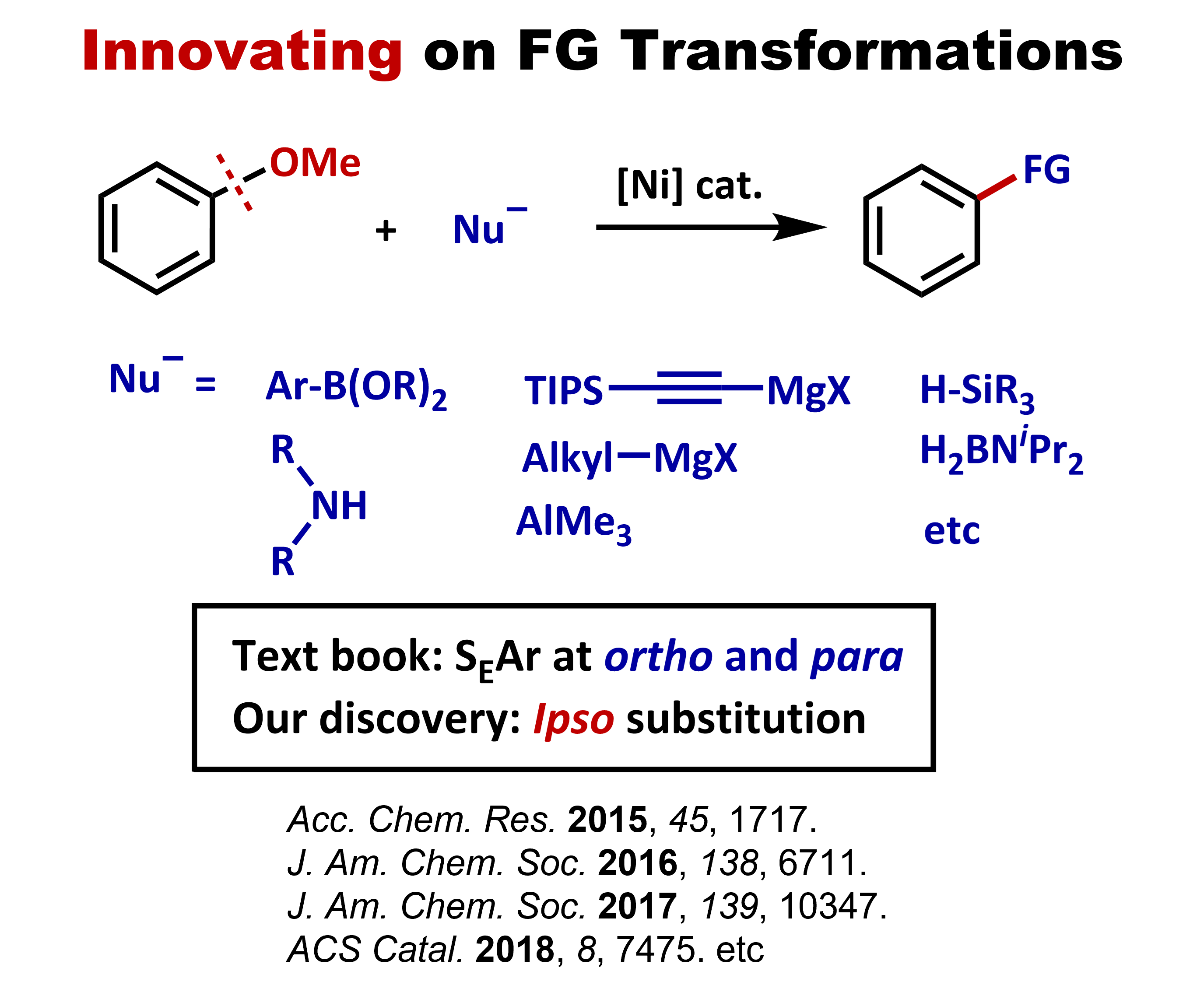
Innovating FG transformation
•
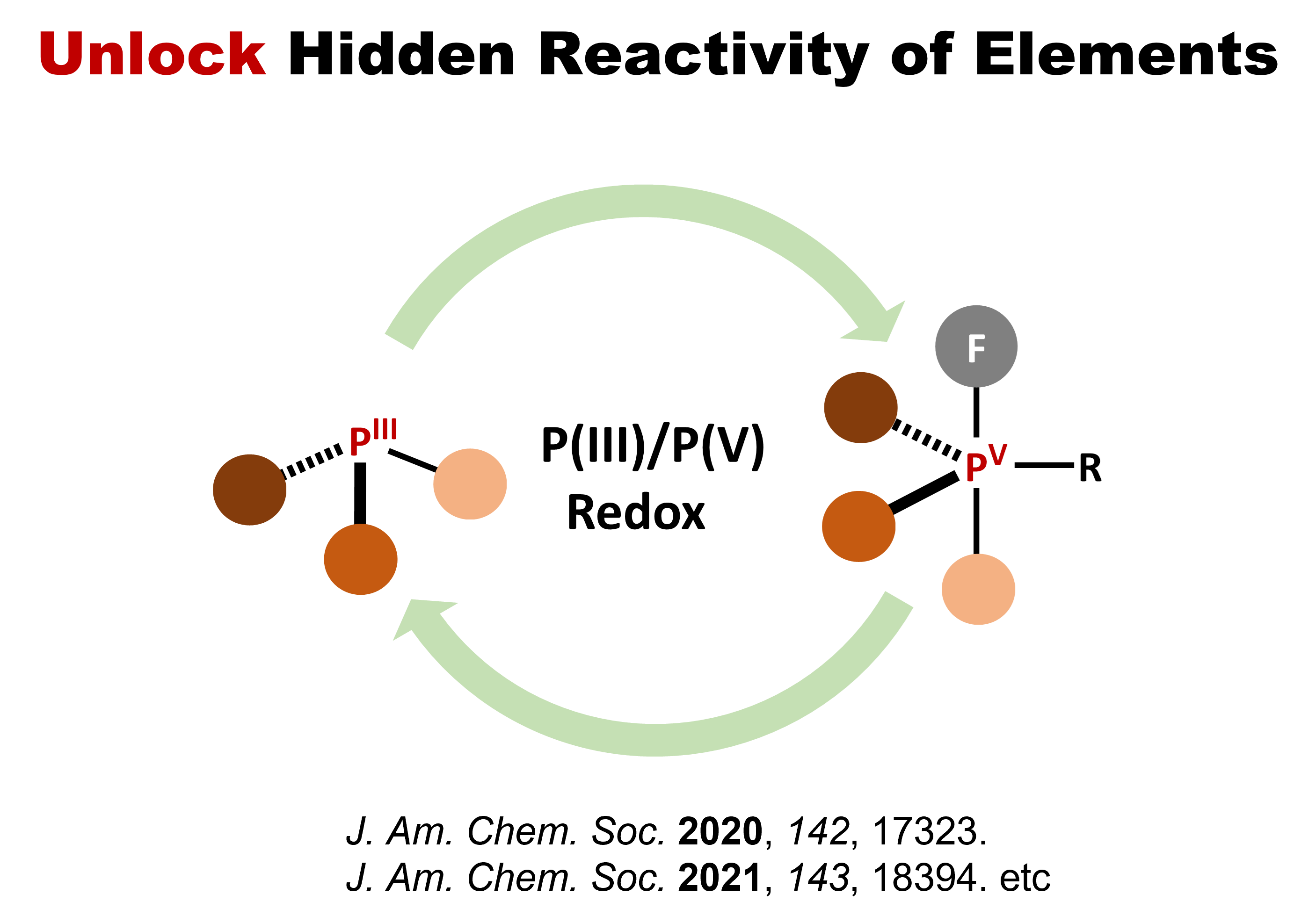
Unlocking hidden character of
elements
Discovering New Chemical Reactions
From pharmaceuticals to liquid crystals, organic
compounds exist everywhere. It is the molecular structure of a compound
that determines its properties and functions. Thus, it is clear that
chemical reactions play an essential role in the creation of new
functional molecules as a means for constructing sophisticated molecular
architectures. Organic chemistry textbooks have taught that a
significant number of chemical reactions have already been developed to
date. Are there any reasons for us to develop new reactions? The answer
should be YES. The emergence of new chemical reactions that, not only
replace existing chemical processes with more environmentally-benign and
energy-saving ones, but also lead to the assembly of molecules that are
otherwise inaccessible continue to be needed. The importance of this
research was recognized by the awarding of the Nobel Prize in chemistry,
even in the 21st century, for research leading to new chemical
reactions, such as asymmetric hydrogenation and oxidation, metathesis,
and cross-coupling. These new reactions have enabled the synthesis of a
series of chiral molecules, polymers and ?-conjugated compounds that had
never existed.
The research objectives of our group include the
development of such new chemical reactions. Our policy is to explore
truly unknown reactivities, rather than modifying or improving existing
methods. Our goal is to discover reactions whose mechanism cannot be
understood at first glance. In other words, we are attempting to explore
reactions out of the realm of current reaction theories. The discovery
of new chemical reactions has the potential to revolutionize our
society, which motivates us to pursue these challenging projects in our
laboratory.
Designing New Reactions
We employ transition-metal complexes as a reliable
tool for inventing new chemical reactions. The introduction of
transition-metal catalyzed reactions, as represented by cross-coupling
and metathesis, during the past several decades have resulted in a
tremendous advance in the field of chemical synthesis. However, it
should be noted that there is still much room for discovering new
reactivities of transition-metal complexes, since a virtually unlimited
number of catalysts can be produced by a combination of a number of
transition metals and a diverse array of ligands, and their reactivities
promise to be infinite. Unfortunately, the theories for metal-catalyzed
reactions are not sufficiently matured to permit the complete design of
a new reaction and to predict the outcome. However, this infancy allows
for experimentalists to approach such issues using their creativity and
imagination. We have attempted to design catalysts, ligands, substrates
and reagents by building a rather risky yet rewarding hypothesis. We
also designed catalytic cycles by designing a balanced combination of
elementary steps that shape the catalytic reactions. Designing a new
elementary step is also a subject of interest in our group. These
designs frequently do not work as well as expected. However, new
reactions can eventually be developed by continuous efforts to polish
the reaction designs or to completely revise the design of such
processes by virtue of serendipitous findings.
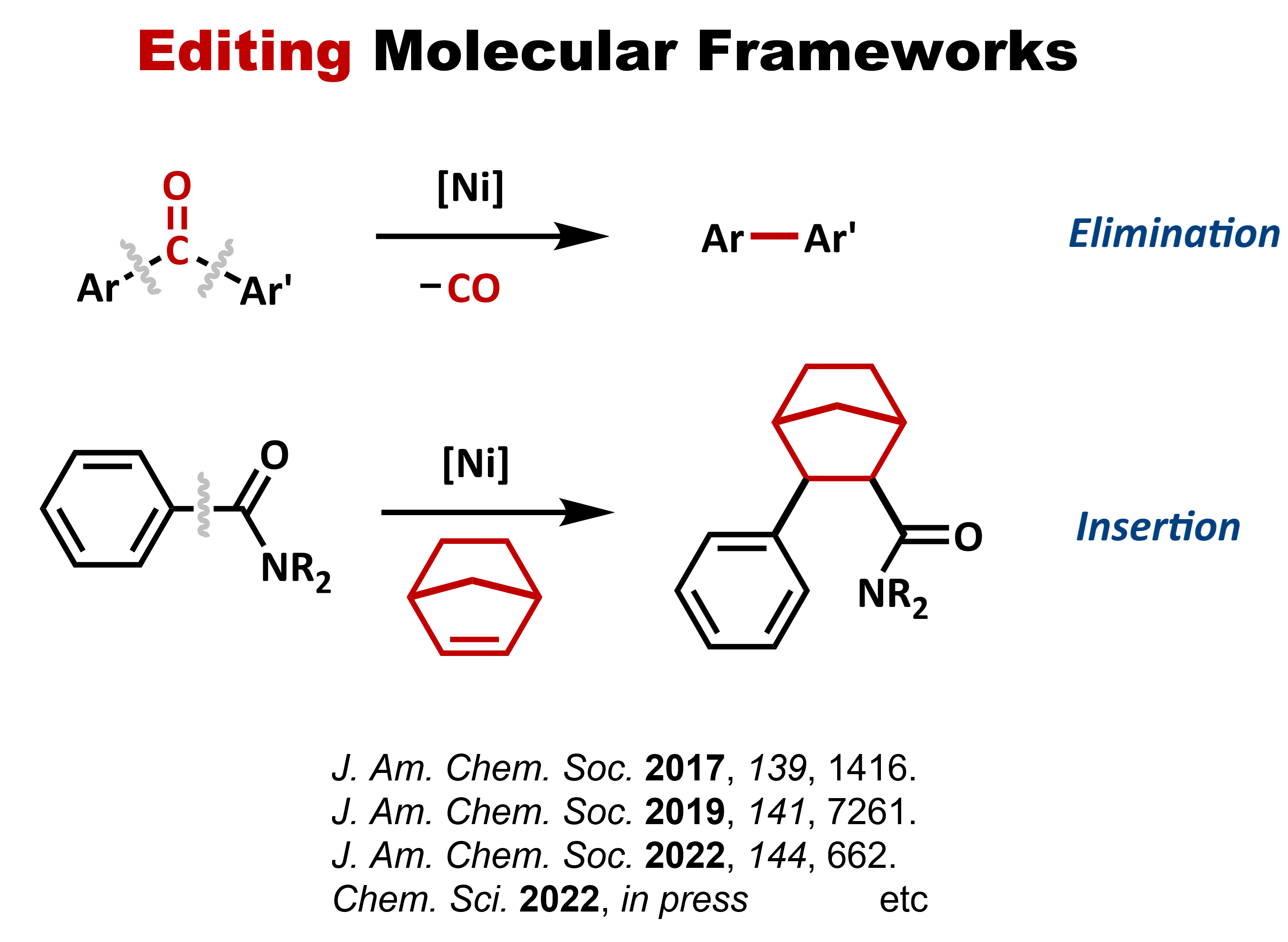
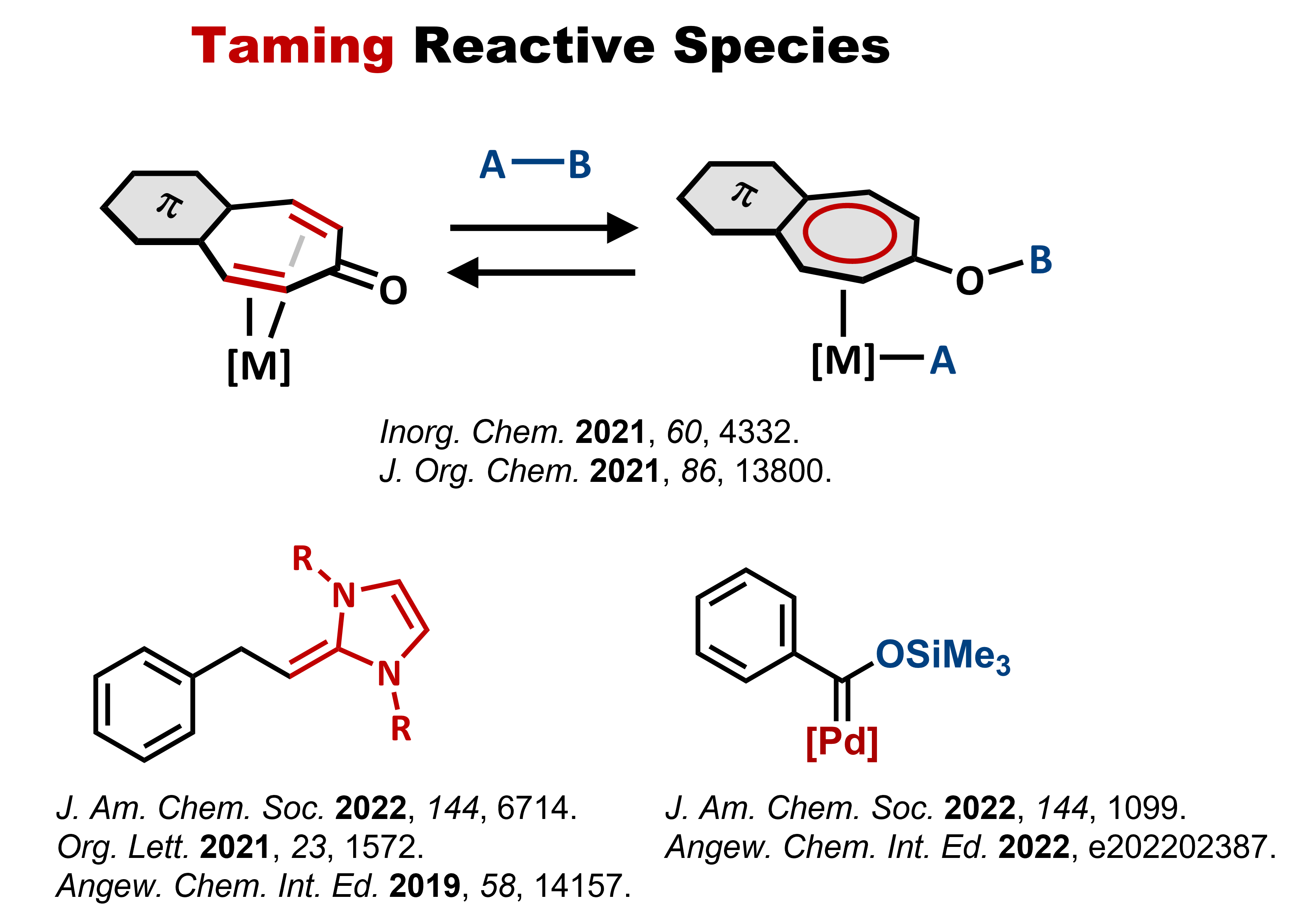
Representative new reactions and molecules that have been discovered to
date are shown below:
•
Innovating FG transformation
• Unlocking hidden character of elements
Copyright(c)All right reserved. The Tobisu Group.





